A self‑adhesive nasal patch delivers L-dopa steadily to the brain while reducing oxidative stress, addressing key limitations of current Parkinson’s treatments.
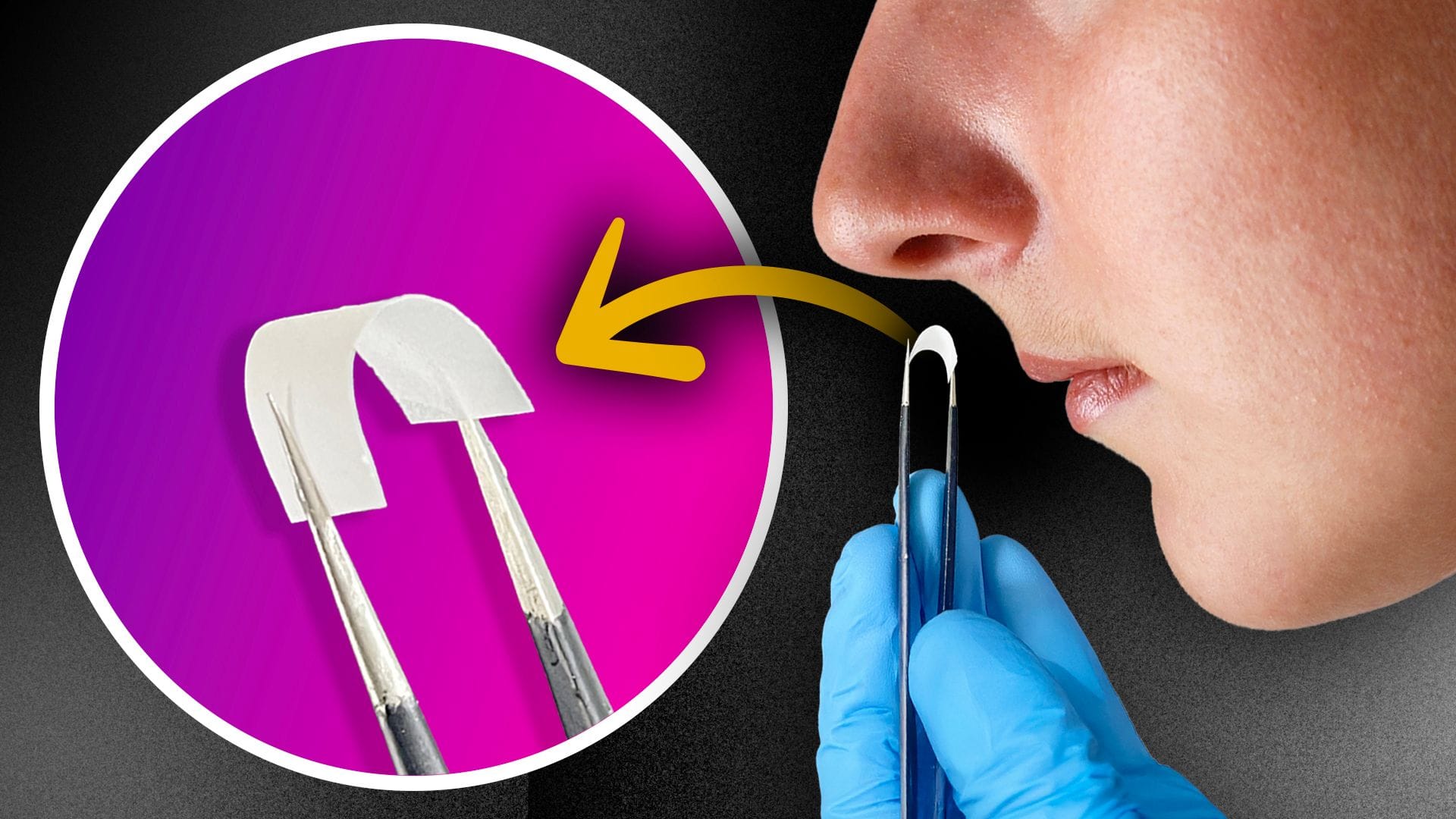
In a new ACS Nano study, scientists report a soft, self‑adhesive intranasal patch designed to deliver levodopa (L-dopa) steadily to the brain while also reducing oxidative stress implicated in Parkinson’s disease progression.
Learn how the patch works in the latest Headline Science video:
The challenge of treating Parkinson’s disease
Parkinson’s disease is one of the fastest‑growing causes of disability worldwide. Motor symptoms such as tremor, stiffness, and slowed movement are the most visible signs of the disease, though non‑motor symptoms are also common. At a biological level, Parkinson’s is marked by the loss of dopaminergic neurons, with oxidative stress playing a central role in neuronal damage.
Treatment still relies primarily on oral L-dopa, which the brain converts into dopamine. But oral dosing poses two persistent challenges: limited bioavailability in the brain and fluctuating drug levels that can trigger periods when symptoms return.
The pros and cons of intranasal delivery
Intranasal approaches aim to deliver drugs more directly to the brain by bypassing the blood–brain barrier. Compounds can travel along olfactory or trigeminal nerve pathways, with some uptake also occurring through the nasal epithelium. But even with intranasal delivery, maintaining stable drug concentrations remains difficult, and most formulations focus solely on dopamine replacement. That leaves oxidative stress, an important contributor to disease progression, largely unaddressed.
Previous studies have shown that nanostructured dopamine formulations delivered intranasally can reach the brain and boost dopamine levels in animal models. However, intermittent dosing still produces concentration peaks and troughs linked to motor complications.
A new strategy
In their recent work, the authors describe a soft, self‑adhesive intranasal patch designed to provide sustained L-dopa delivery while also scavenging reactive oxygen species (ROS). The patch is made from silk fibroin nanofibers that conform to the moist nasal cavity without additional adhesives. Embedded within the fibers are chitosan nanoparticles carrying L-dopa alongside copper oxide nanoparticles, which act as antioxidants. As the silk matrix gradually degrades, both components are released steadily, avoiding abrupt spikes in drug concentration.
In mouse models of Parkinson’s disease, treatment with the patch reduced brain ROS levels by more than 50% and led to improvements in motor performance. Animals receiving the patch traveled farther and completed coordination tasks more quickly than those given oral levodopa or intranasal drops, with movement patterns more closely resembling healthy controls.
These preclinical results show how combining sustained dopamine supplementation with oxidative stress mitigation may address multiple challenges associated with Parkinson’s treatment. While further studies are needed to assess long‑term safety, dosing, and clinical relevance, the work points to a promising intranasal drug‑delivery platform that emphasizes stability, retention, and neuroprotection—key considerations for managing a chronic neurodegenerative disease.
Watch more Headline Science on YouTube!
The video above is brought to you by the ACS Science Communications team. To watch more exciting videos and shorts covering some of the latest research in ACS journals, visit the Headline Science page on YouTube.
Video credits:
Written and edited by Vangie Koonce
Narrated and hosted by Emily Schneider
Series produced by Vangie Koonce, Anne Hylden, Andrew Sobey, Janali Thompson, Elaine Seward, Darren Weaver, and Jefferson Beck
Executive produced by Matthew Radcliff
Research videos from Ting Wang, Ph.D.
Treating Parkinson's: Explore related articles from ACS journals
Probiotic Spore-Based Oral Biotherapeutics Promote Brain Targeting toward Gut-Brain Dual-Regulation for Parkinson’s Disease Treatment
Qianhua Feng, Yiwei Li, Liying Sun, Jia Zhang, Lisha Shou, Jieyu Zhu, Huimin Zhou, Bingqian Si, Xinyu Gu, Chunlei Du, Zhenzhong Zhang, and Lei Wang*
DOI: 10.1021/acsnano.5c17340
Discovery of Potent, Selective, CNS-Penetrant Macrocyclic LRRK2 Inhibitors for the Treatment of Parkinson’s Disease
Elsie C. Yu*, Hua Zhou*, Xin Yan, Kaitlyn M. Logan, Derun Li, Anmol Gulati, Kelsey E. Poremba, Michael J. Ardolino, Jing Su, Dong Xiao et al.
DOI: 10.1021/acs.jmedchem.6c00238
Treatment of Parkinson’s Disease with an Anti-Inflammasome NLRP3 Inhibitor Derived from a Natural Product
Wenqian Li, Jiuliang Zhang, Qiang Chen, Biaobiao Luo, Bo Zhou, Jun Wang, Yang Yang, Yi Liu, Shuixiu Wen, De-Xin Kong, Jian Chen, Shaozhong Wei, Pierre Duez, Avez Sharipov, Yan Li, and Xuebo Hu*
DOI: 10.1021/acsptsci.5c00109
An Artificial Intelligence Olfactory-Based Diagnostic Model for Parkinson’s Disease Using Volatile Organic Compounds from Ear Canal Secretions
Xing Chen, Yi Li, Chenying Pan, Shenda Weng, Xiaoya Xie, Bangjie Zhou, Hao Dong*, and Danhua Zhu*
DOI: 10.1021/acs.analchem.5c00908
Neurotransmitter Dysregulation in Parkinson’s Disease: Pathophysiological Insights and Therapeutic Perspectives
Feyza Sule Aslan, Beyza Barut, Sudenaz Ozturk, Tugba Eyigurbuz, and Enes Akyuz*
DOI: 10.1021/acschemneuro.5c00809
Advancing Parkinson’s Prevention: Brain-Specific Lipopolyplex Delivery of the GDNF as a Neuroprotective Gene Therapy
Yujing Zhang, Yujing Huang, Zhonghua Lu, and Zhen Yuan*
DOI: 10.1021/acsmaterialsau.6c00024